 I.
I.
In 1999, pharmaceutical giant Bristol-Myers Squibb struck a hell of a deal.
They reached an agreement with a Japanese pharmaceutical company, Otsuka, to market one of their products in the United States. Under the terms of the agreement, Bristol-Myers would help lead the clinical trials to get the new medication approved by the U.S. Food & Drug Administration (FDA). In return, Bristol-Myers would get 65% of the sales of the medication in the United States.
The medication was Abilify (aripiprazole).
Discovered in 1988, Abilify was part of a new class of medications called atypical antipsychotics. These ‘atypicals’ promised to revolutionize the care for patients with schizophrenia. But Abilify seemed especially promising. Pharmacologically, Abilify didn’t bind to histamine receptors (resulting in less sedation) and served as a partial agonist of dopamine receptors (theoretically leading to more stable treatment effects and fewer extrapyramidal symptoms). There was real optimism that Abilify would prove to be the ‘best in class’ medication, and Bristol-Myers Squibb was getting on the ground floor.
Still, there was a problem: Bristol-Myers had been beaten to market by its competitors.
Johnson & Johnson (Risperdal), Eli Lilly (Zyprexa), and AstraZeneca (Seroquel) had already received FDA approval for treating adults with schizophrenia. Many of the patients that might have been good candidates for Abilify were already receiving a competitor’s product.
However, their competitors’ head start did give Bristol-Myers Squibb something useful: information.
The company knew that some doctors had been prescribing atypical antipsychotics “off label” in difficult-to-manage conditions such as pediatric mood disorders or dementia-related psychosis. There weren’t studies to prove that these medications were safe and effective in such scenarios… but the physicians treating these patients didn’t have many other options. Why not try Abilify?
And so, rather than fighting with their competitors for a bigger slice of a relatively-small pie, Bristol-Myers Squibb saw the opportunity make the pie bigger by bringing Abilify into a new market.
–
II.
The Comprehensive Osteopathic Medical Licensing Examination of the United States (COMLEX-USA) is a three-part test taken by osteopathic physicians.
The purpose of COMLEX-USA is to ensure that osteopathic physicians (DOs) possess the requisite knowledge and skills to receive a medical license. Toward that end, the test is criterion-referenced and specifically designed to discriminate between an examinee with borderline proficiency and one who is incompetent. (Even though the National Board of Osteopathic Medical Examiners (NBOME) report a numeric score for their Level 1, Level 2 CE, and Level 3 exams, it’s not entirely clear what numerical differences in score above the passing standard really mean.)
However, the COMLEX-USA isn’t the only licensing examination that DOs can take.
Nowadays, in addition to the COMLEX-USA, most DOs take the United States Medical Licensing Examination (USMLE). Unlike MDs, who are required to take the USMLE, DO students do so voluntarily – in the hopes of bettering their applications with residency program directors who are more familiar with the USMLE than COMLEX.
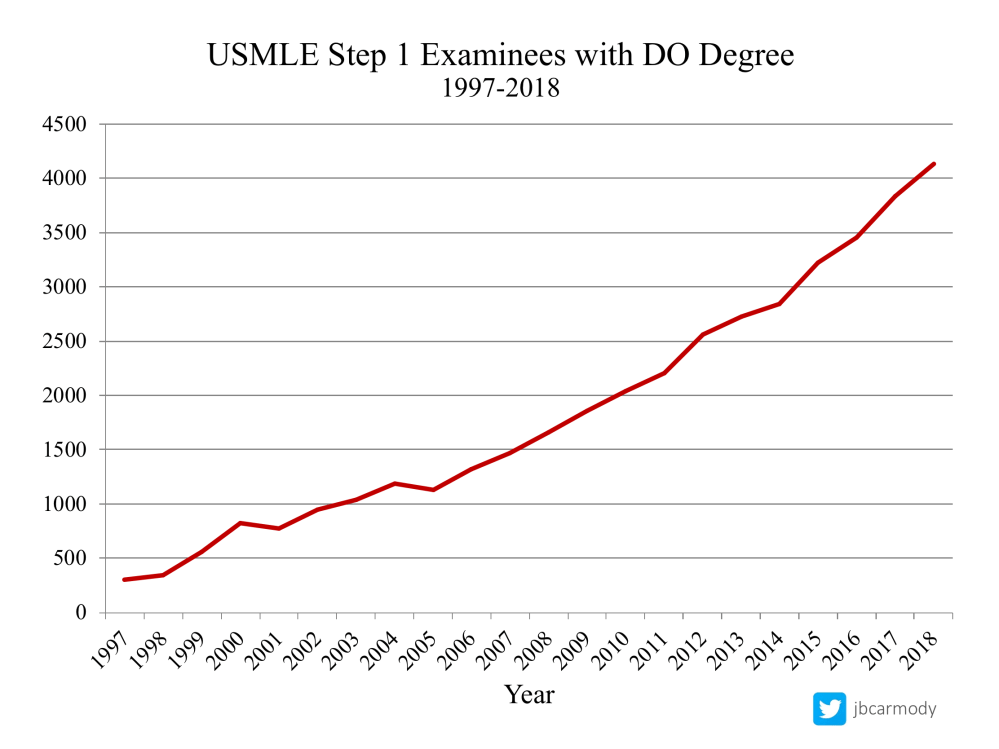
A large and growing number of osteopathic medical students are taking the USMLE.
But even though most DOs aren’t taking the USMLE for licensure, they could: almost all states allow DOs to become licensed by completing the USMLE series.*
Historically, few DOs have pursued this option. There was little reason to.
Osteopathic medical schools require their students to take the initial parts of COMLEX to graduate. Once you’ve taken Level 1, Level 2-CE, and Level 2-PE, only the Level 3 remains to receive a full medical license.
On the other hand, completing the USMLE series would require DO students to take USMLE Step 2 CS – a $1600 pass/fail exam that residency program directors don’t care about and that one in twenty students fails – as well as USMLE Step 3. Why choose the riskier and more expensive path?
–
III.
To expand the market for Abilify, Bristol-Myers Squibb created new teams of sales representatives to call upon pediatric psychiatrists and physicians who worked in nursing homes and long-term care facilities.
These physicians didn’t care for many adults with schizophrenia. Of course, that was kind of the point.
[Bristol-Myers sales representatives] called on pediatric doctors almost nonstop since 2002 and “sold symptoms” such as agitation, a feature of both schizophrenia and other conditions found in juveniles, including attention deficit hyperactivity disorder (ADHD) and oppositional defiant disorder. In a pitch to adolescent providers, reps would ask “if they had female patients who … can’t concentrate in school, are moody, and are preoccupied with their weight.” Abilify was then proposed as a solution to these ills.
At nursing homes and long-term care facilities, reps sold agitation and lethargy, telling medical directors that Abilify would help them to manage their patients’ behavior. One pitch suggested the drug would result in fewer calls from staff in the middle of the night to voice complaints of unruly residents. . . As instructed in their training, the reps asserted that Abilify was “not contraindicated” in the elderly (the lawsuit says geriatric use had not been studied), and if akathisia or other side effects were a concern, then the doctors should “start low, go slow,” break the tablets in half, or even drop the liquid form of Abilify into a resident’s juice.
-Matt Hrodey, “The Selling of Abilify”
The sales pitches worked.
By 2008, Bristol-Myers Squibb took in approximately $2 billion in revenue from Abilify.
–
IV.
Among many other things, COVID-19 changed medical licensure exams. Or, at least it did for some test-takers.
The National Board of Medical Examiners (NBME) committed to a long-term suspension of the USMLE Step 2 CS exam. The NBME’s announcement made it possible for students to go on with their lives and for medical schools and licensing authorities to make workarounds for a world without Step 2 CS. (So far, the exam hasn’t been missed.)
In contrast, although students have been unable to take the COMLEX Level 2-PE since March 2020, the NBOME has refused to commit to a long-term test suspension. Instead, they’ve doled out short-term updates on their website, each month promising that the Level 2-PE exam will be back soon.
The NBOME’s unwillingness to concede that maybe, just maybe, requiring thousands of students from around the country to gather for an in-person standardized patient exam wasn’t a good idea in the year 2020 has left students in limbo.
Even though the NBOME begrudgingly admitted that it might be impossible to test all of the fourth-year DO students before next spring – meaning that class of 2021 may be permitted to graduate without taking Level 2-PE – they have steadfastly insisted that all DOs will have to take the exam at some point.
COMLEX-USA Level 2-PE will still need to be completed for full licensure, and is considered a necessary and ongoing requirement for the licensure of osteopathic physicians in all 50 states.
–Joint Statement from AACOM, AOA, COCA, and NBOME (emphasis added)
This position is light on logic, but strong on business protectionism.
Yeah, it may be illogical to force an intern to leave the ICU or hospital ward, where they were providing real care for real patients to travel to Conshohocken, Pennsylvania to participate in a $1295 doctor cosplay to prove that s/he is qualified to do the thing that s/he was already doing.
But from a business standpoint, it makes perfect sense. If you allow some DOs to become licensed without COMLEX Level 2-PE, it is going to be very difficult to in the future to articulate a convincing reason why students should again be required to take the test.
On the other hand, until USMLE Step 2 CS returns – and at some point, it will – qualified examinees can register for USMLE Step 3. That means that a DO graduate who took USMLE Step 1 and Step 2 CK for residency selection could become eligible for licensure by taking just one last computer-based test, avoiding the expensive clinical skills exams altogether.
Suddenly, the USMLE pathway to licensure was starting to look more appealing to DO students.
–
V.
Despite booming sales, evidence was mounting that off-label use of Abilify and other atypical antipsychotics might not be a good idea.
It turned out that elderly patients treated with Abilify were more likely to develop adverse effects like somnolence, lethargy, difficulty swallowing, and increased salivation – a toxic combination that increased the risk of aspiration.
Similarly, children and adolescents with depression who were treated with Abilify had emergence of suicidal thoughts and behaviors more often than expected. For every 1000 patients <18 treated with Abilify instead of placebo, there were 14 additional cases of suicidality (versus 1 fewer case in adults 25-64).
Soon, the FDA slapped Abilify with a “black box” warning – the strongest warning the agency can give, used to highlight potentially life-threatening adverse effects.

The Abilify black box warning, from the package insert.
–
VI.
In August, the National Resident Matching Program (NRMP) issued their biennial Program Director (PD) Survey.
Soon thereafter, the NBOME Tweeted this:

The NBOME’s Tweet.
There are at least two big problems with this Tweet.
The first is its basic truthfulness.
The 86% figure triumphantly touted in the Tweet above doesn’t actually appear in the NRMP report.
It’s derived from Figure 4, which shows that only 45% of programs use specific COMLEX Level 1 scores to screen applicants, while another 41% require applicants to pass COMLEX Level 1 in order to be considered for an interview.
Combining these two figures is a bit curious, especially when the first item on the NRMP survey specifically provides the data suggested by the NBOME’s Tweet.
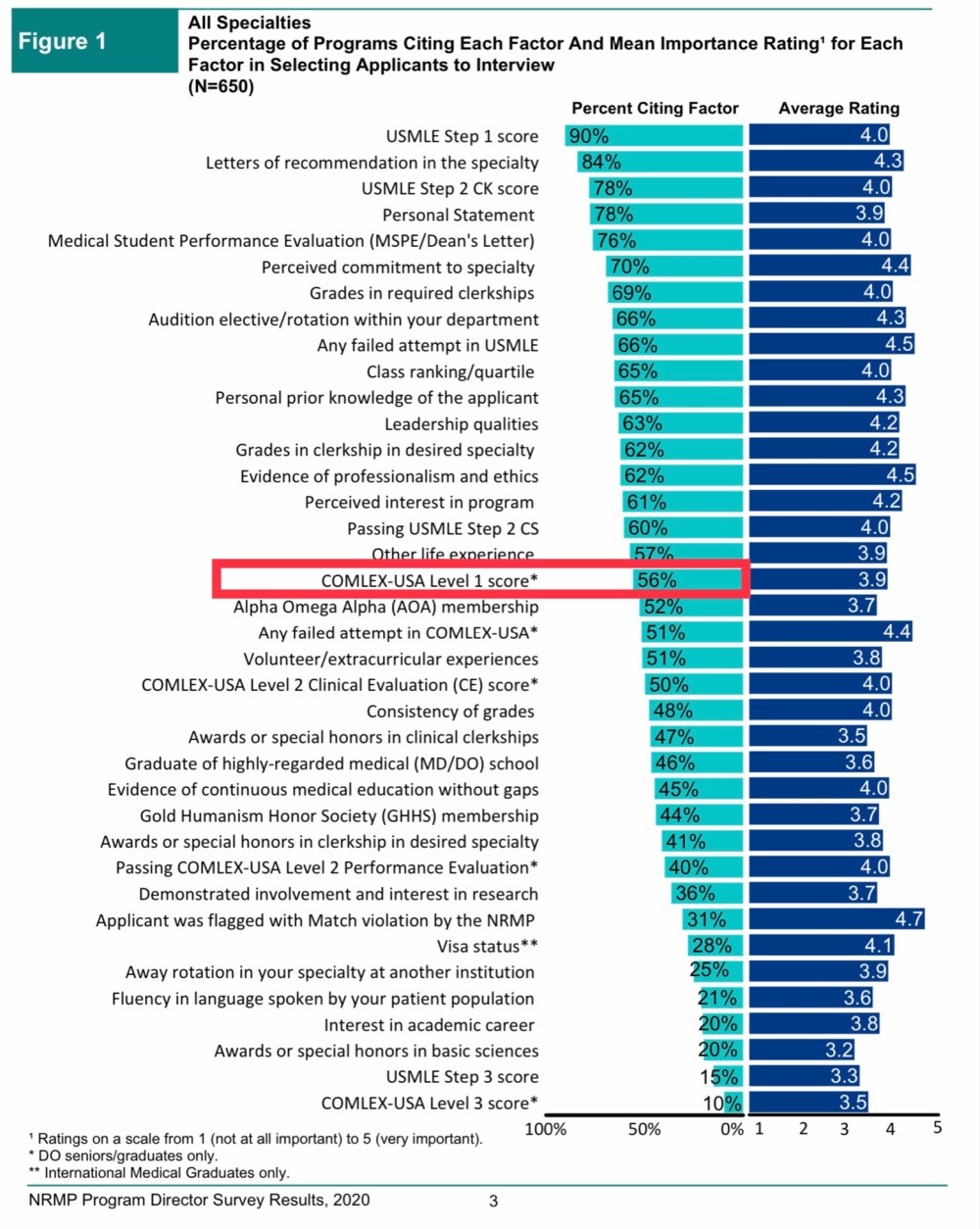
When does 56% equal 86%? When the NBOME says it does.
But in my opinion, there’s an even bigger problem with the NBOME’s Tweet.
–
VII.
It’s important to note that the physicians who prescribed Abilify ‘off-label’ didn’t break any laws. Real life medical practice can be messy. Doctors often have to make therapeutic decisions using their best judgment, in the absence of high-quality evidence.
In contrast, after the FDA approves a medication for the treatment of a specific condition, the company that makes the medication is expressly forbidden from marketing their products for purposes for which they have not been proven beneficial.
There’s a reason for that. While a physician has to act on what is in the best interest of their patient, the pharmaceutical company will act in the best interest of their financial bottom line.
By encouraging doctors to use Abilify for purposes for which it had not been proven safe and effective, Bristol-Myers ran afoul of the False Claims Act – and attracted the attention of federal prosecutors.
In 2007, Bristol-Myers settled with the U.S. Department of Justice for over half a billion dollars.
–
IX.
Is there a parallel between pharmaceutical companies pushing off-label use of their products and the NBOME marketing the COMLEX for a purpose for which it was never intended (and likely functions poorly)?
I say there is.
For the NBOME, selling COMLEX-USA is a gainful enterprise. Their CEO received total compensation of $606,214 in 2018 (including $30,679 in bonus and incentive compensation). Let’s just say they have some skin in the game.
If residency program directors want to use COMLEX-USA scores, well, caveat emptor. But the NBOME has an ethical obligation (and you’d hope, enough good sense) to refrain from clownishly promoting unproven uses of their products – especially when doing so requires fudging data that are easily fact-checked.
–
X.
In February 2020, right after the USMLE announced that results of their Step 1 exam would be soon be reported pass/fail, I talked with Someone In the Know who said COMLEX-USA would soon follow suit. According to this person, it was just a matter of time; they just needed a few more people to sign off on the decision.
But time went by – and nothing happened.
The NBOME did acknowledge that they were considering score reporting changes, and stated that more information would be available “as early as July 2020.”
Will the NBOME make COMLEX-USA Level 1 pass/fail? Or will they double down on a scored test, in the hopes that doing so will maintain the importance of their signature product? Even if they do eventually announce a pass/fail Level 1 exam, their delay in doing so speaks to the presence of significant internal debate on this issue.
One thing is certain: recent trends threaten the NBOME’s very existence.
A growing number of DO students have real animosity toward the NBOME. They view the NBOME as a protection racket, and consider their tone-deaf and self-interested refusal to suspend the Level 2 PE as nothing less than financial predation. They bristle at the de facto requirement to take both the USMLE and COMLEX – and increasingly view the latter as being the superfluous exam.
For now, the NBOME is secure. They enjoy the full support of the American Osteopathic Association (AOA) and their Commission on Osteopathic College Accreditation… and so long as DO schools are required to make students take COMLEX as an accreditation standard, the NBOME’s business is safe.
Trouble is, contemporary DO students don’t seem to feel much allegiance to the AOA, either. Many view the AOA’s insistence on protecting the ‘distinctiveness’ of osteopathic medicine as a hindrance to their own careers.
But it won’t be long until the current generation of DO students outnumber their predecessors. There are now 37 colleges of osteopathic medicine in the United States. Fifty years ago, there were five. If expansion continues, more new DOs will graduate each year than there were total DOs practicing in post-WWII America.
And as this generation of DOs “grows up” and takes on leadership in the AOA, it’s easy to imagine that the organization’s willingness to prop up the NBOME will dwindle.
From the outside, the NBOME has the look of an organization in disarray. They realize that they are in an existential struggle – but can’t quite reckon how to get out of it.
Well, I have some ideas. And in the rest of this series, I’m going to show what the NBOME is doing wrong – and what they ought to do to better serve osteopathic physicians and the public.
–
* – State requirements for medical licensure on available on the Federation of State Medical Boards website. The American Osteopathic Association compiles a similar list, which was last updated in May 2020. California, Florida, Michigan, Pennsylvania, and West Virginia are the only states noted as requiring the COMLEX for initial DO licensure. [ADDENDUM: Interestingly, the AOA seems to have password protected their list after it started getting attention. However, it’s archived on the Wayback Machine for anyone interested.]
–
CLICK HERE FOR PART 2
–
YOU MIGHT ALSO LIKE:
Journal Club: COMLEX-USA and Physician Discipline
Grand Rounds: The Rise and Fall of USMLE Step 1